
Practical limits to polydispersity for quality colloidal crystals
An experimental approach to systematically explore the link between particle polydispersity and the structure thin-film colloidal crystals.
Over the last few years, most of us have used hand sanitisers from a variety of brands and likely noticed that each formulation offered a distinct hand feel. Have you come across sticky hand sanitisers, or ones that do not spread nicely? Rheology, the branch of physics that deals with the deformation and flow of matter, can explain why this happens and help overcome these issues.
In work published in Rheologica Acta, researchers from the Edinburgh Complex Fluids Partnership (ECFP) set out four principles for designing the rheology of thickened alcohol-based hand rubs with acceptable handleability and hand feel based on the soft-matter science of the system. The criteria chosen were low run off, spreadability, smoothness, and non-stickiness.
While this work focused on hand sanitisers, the general approach can be applied to a variety of different systems and demonstrates how soft matter physics can be used to create a framework to guide the design of formulations with desirable properties.

To avoid wasted product and ensure effective microbial decontamination, hand sanitisers should ideally be thick enough to stop the solution from running off users’ hands. This property relates to the yield stress of the sample – the stress at which it will begin to flow – and/or its viscosity. When we apply sanitiser and start to move our palms to rub it in, gravity acts on the product and can exceed the yield stress and induce run off. The ECFP team therefore calculated the approximate yield stress of a hand sanitiser gel that would prevent this from happening. Additionally, in the moments after depositing sanitiser onto our hands before rubbing it in, a product without yield stress but with higher viscosity will have a slower run-off speed. Accordingly, ECFP researchers also determined the ideal viscosity of a hand sanitiser to ensure low run-off and practical handleability.
Products like hand cream and hand sanitisers that are rubbed rapidly on to the skin normally shear thin, i.e. their viscosity decreases as the applied deformation increases. The degree of shear thinning needed is dictated by the desired viscosity during the rubbing process. Through approximating a reasonable hand rubbing speed, and based on literature reports of what consumers consider as good spreadability, the ECFP team determined the ideal shear thinning behaviour of a hand sanitiser to optimise its hand feel for users.
Hand sanitisers are often thickened through the addition of polymers, and in turn, they can become elastic. In soft matter physics terms, this creates normal stress differences on the formulation. In the latter stages of rubbing, this force prevents abrupt skin-to-skin contact, and previous studies have suggested this is what makes such formulations feel smooth during use. As the third of their design criteria, ECFP researchers thus proposed that hand sanitisers have a measureable normal stress difference.
Sanitisers are commonly used in “on the go” situations and it is not desirable for the product to leave users’ hands feeling sticky. Such stickiness results from the solution forming tiny threads between two distinct skin surfaces (for example, a finger and palm) that take a noticeably long time to break. This behaviour is related to the extensional rheology of the sample. As the name suggests, extensional rheology reflects how a material responds as it is stretched. To prevent stickiness, the team determined that any filamentous threads formed during hand sanitiser use should ideally break in around one second or less.
Using the above design criteria, the ECFP team undertook experimental evaluations of a commercial hand gel (Purell) and a variety of simplified formulations thickened with four different microgel and linear polymer additives.
Run off, spreadability, and smoothness were investigated using steady shear rheology. A DHR-2 rheometer was used to measure the viscosity, stress and first normal stress difference for each sample and the yield stress values were obtained. The results showed that all four additives fitted the design criteria for spreadability, three met the criteria for smoothness, and all but the most dilute linear polymer solution offered low run-off.
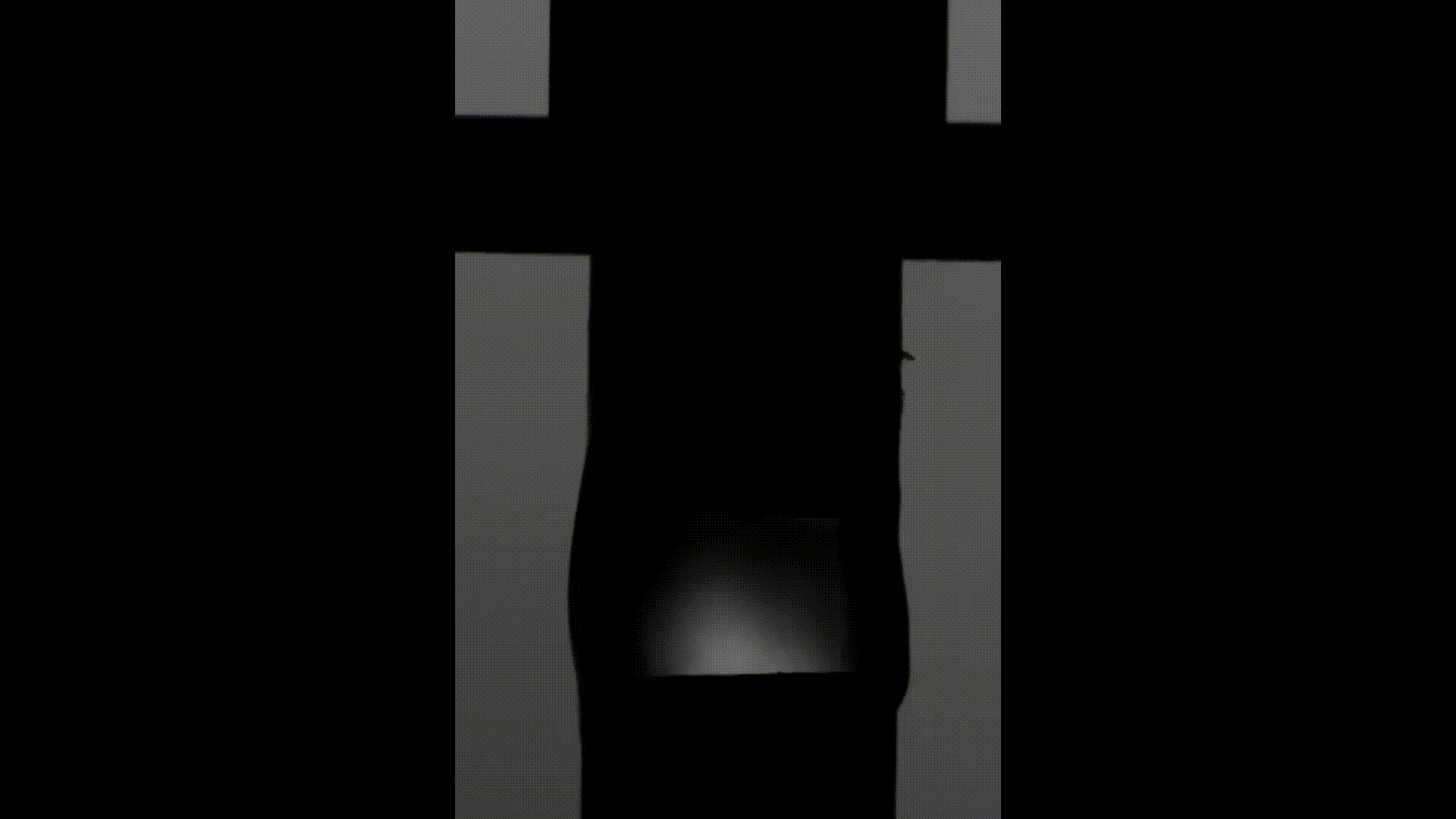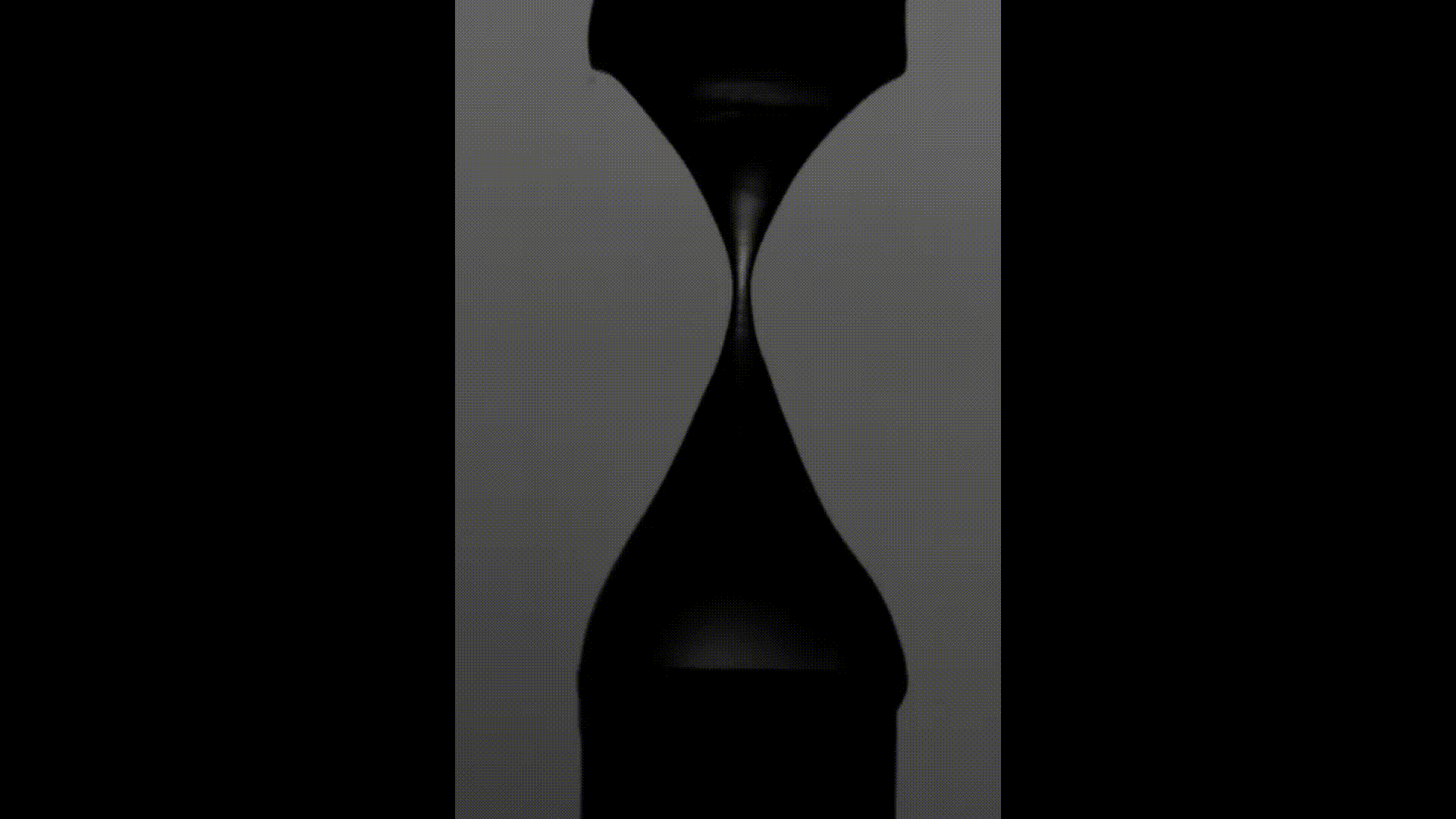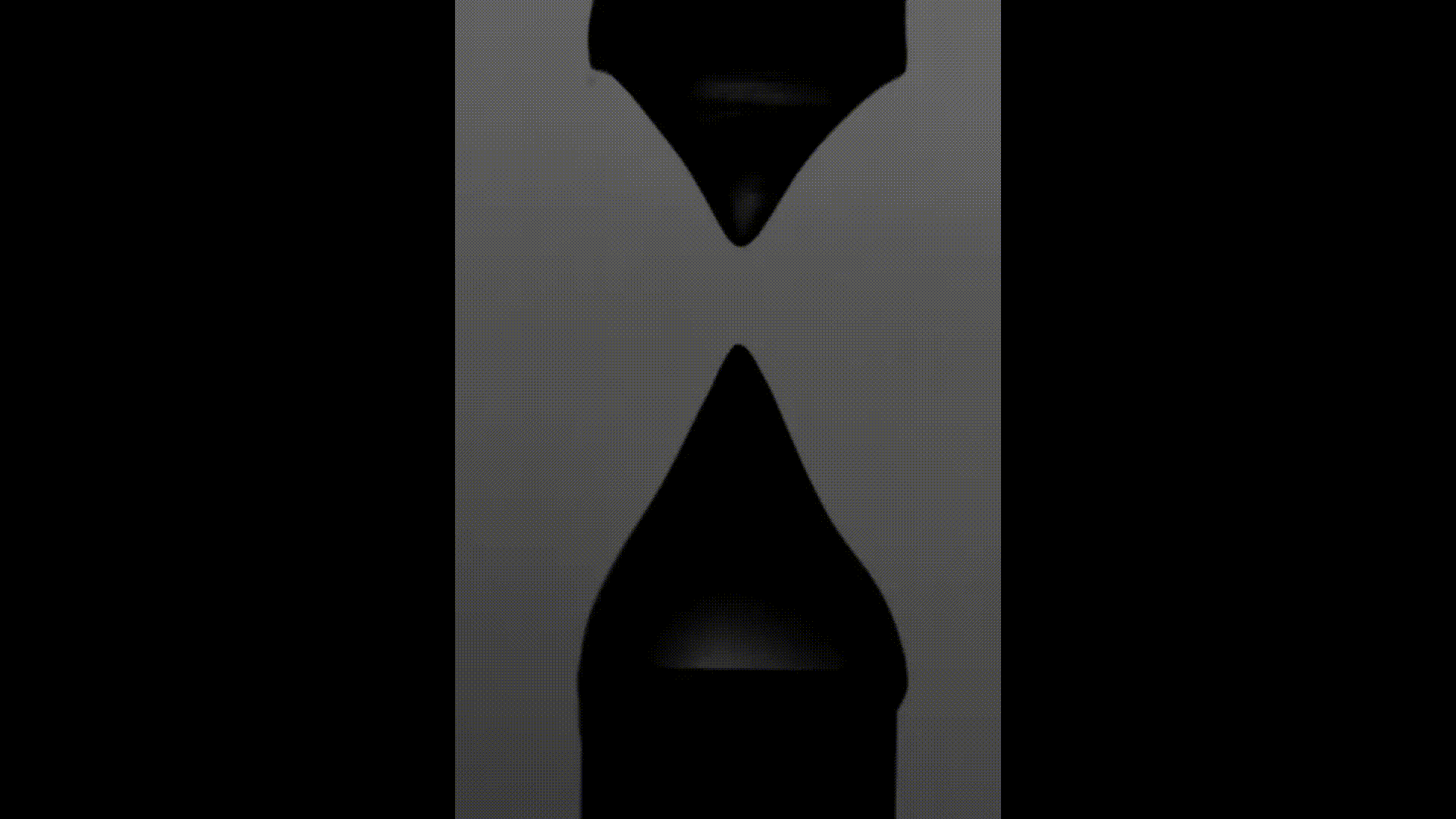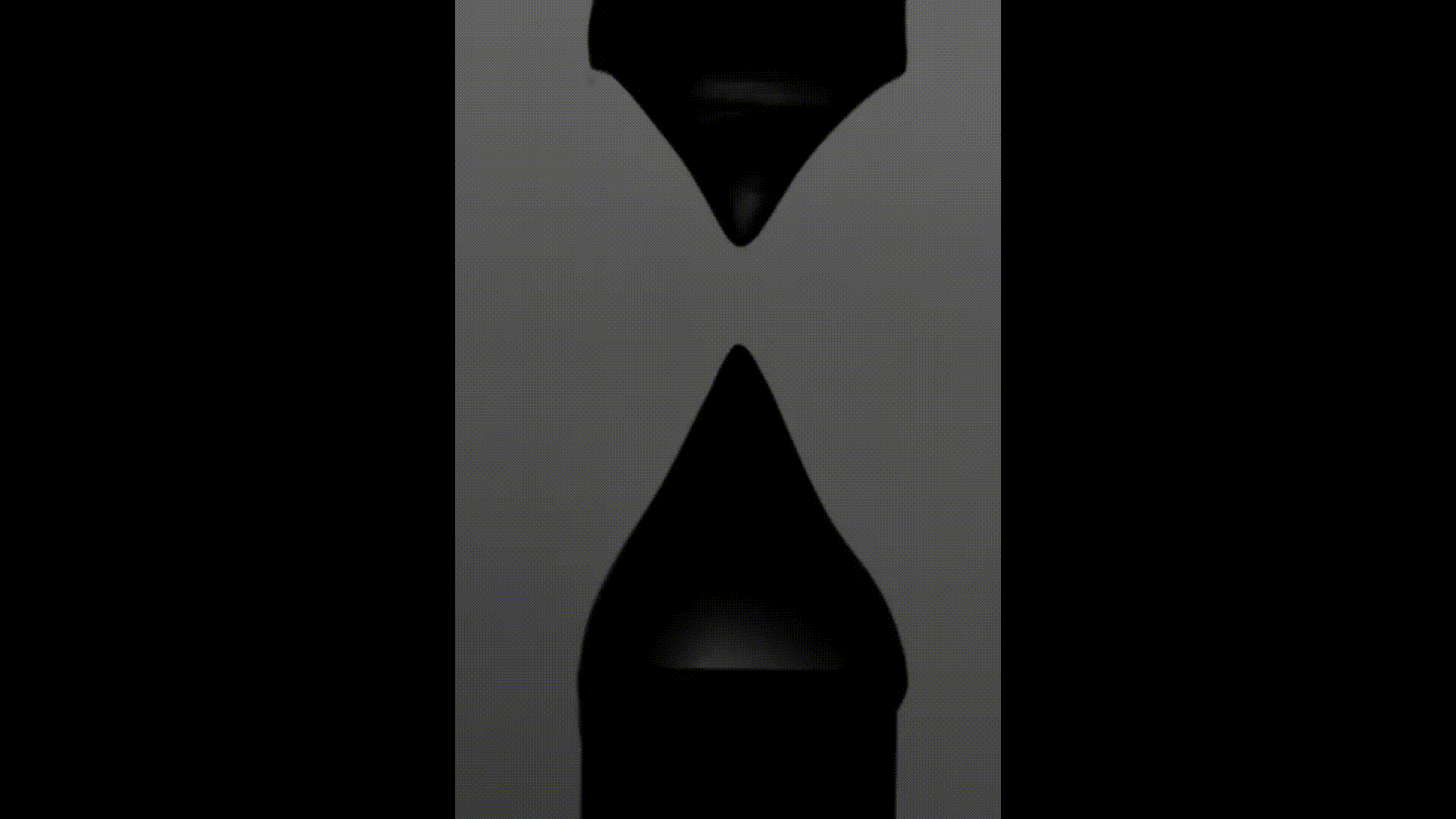
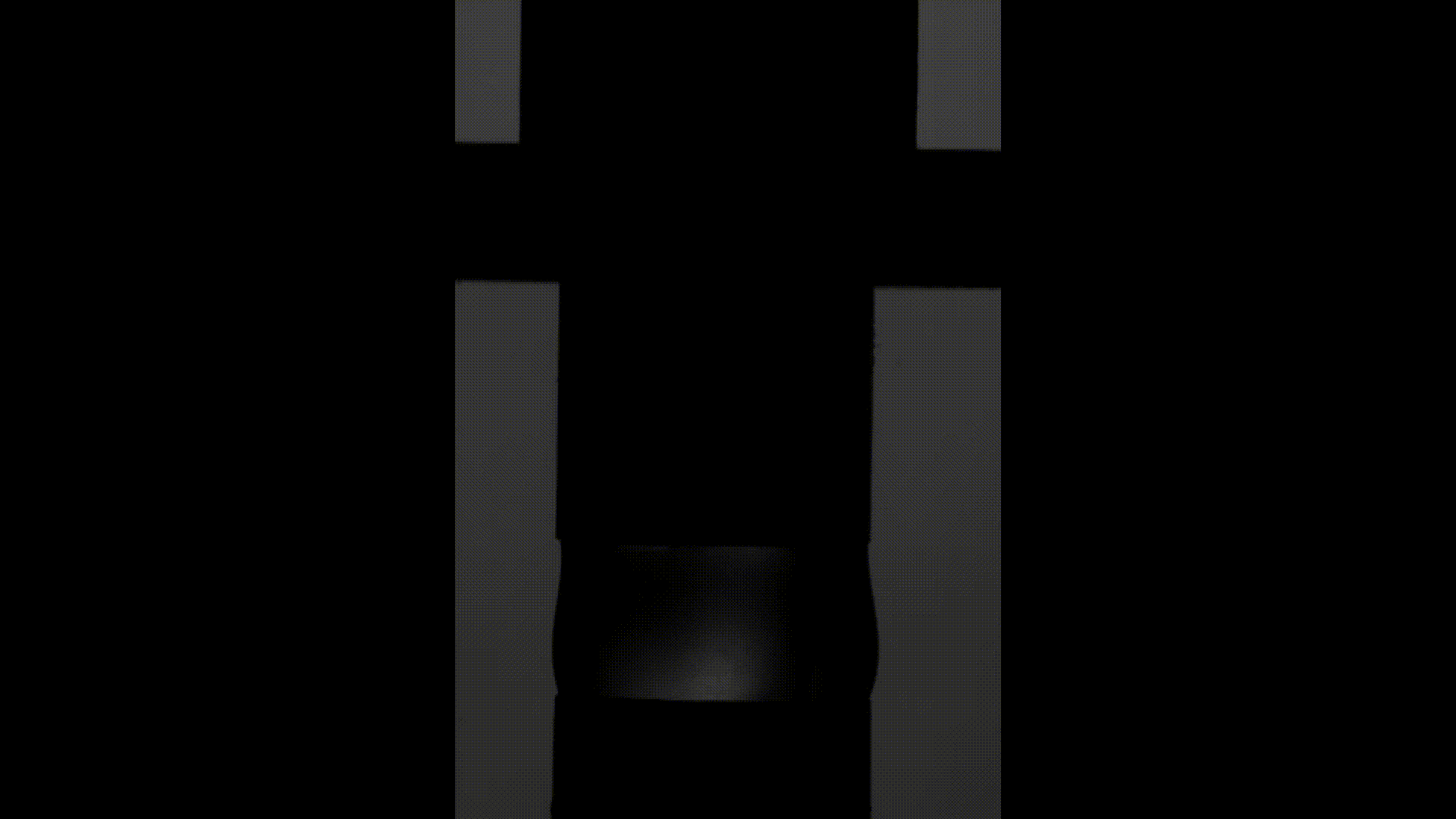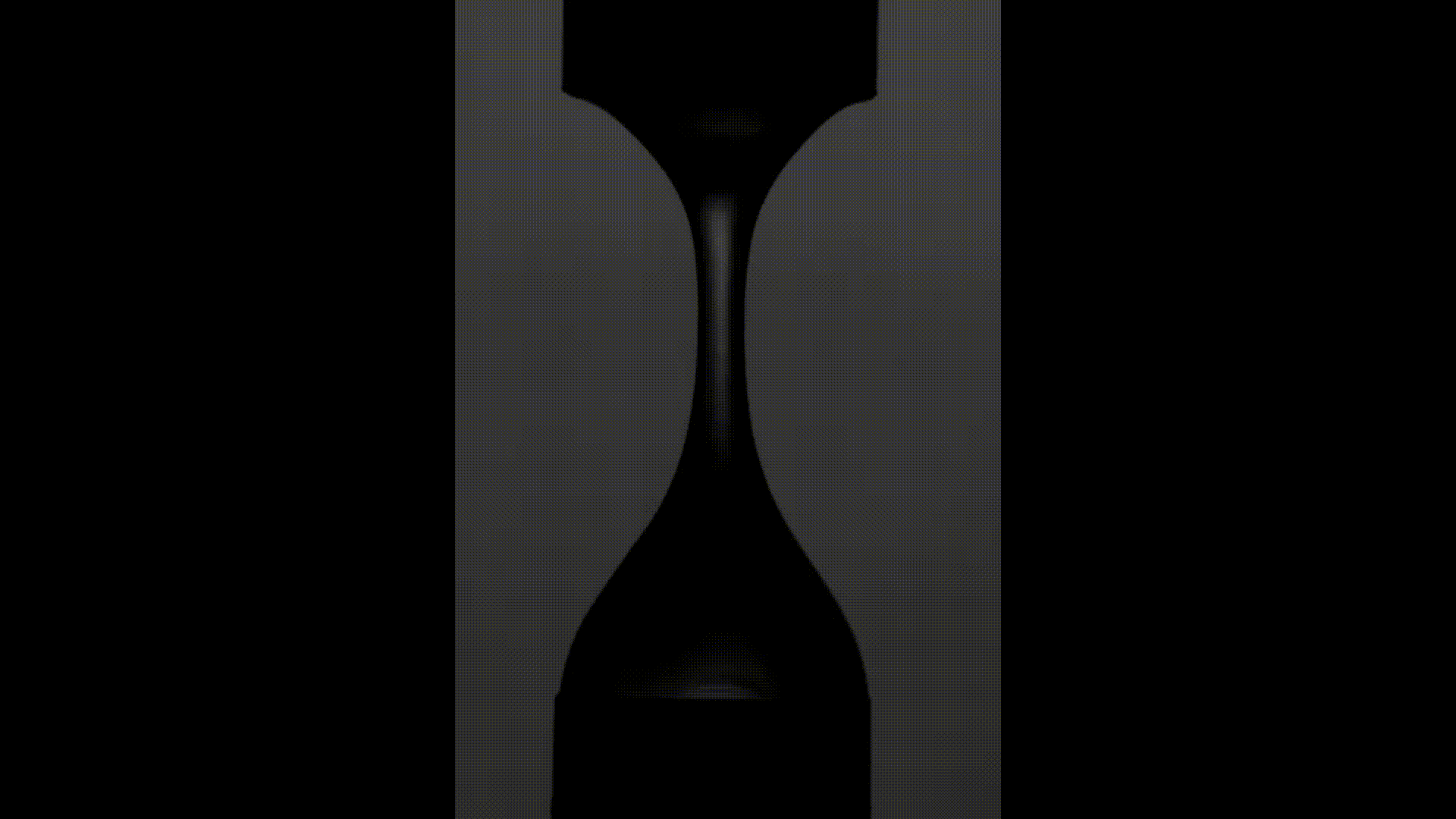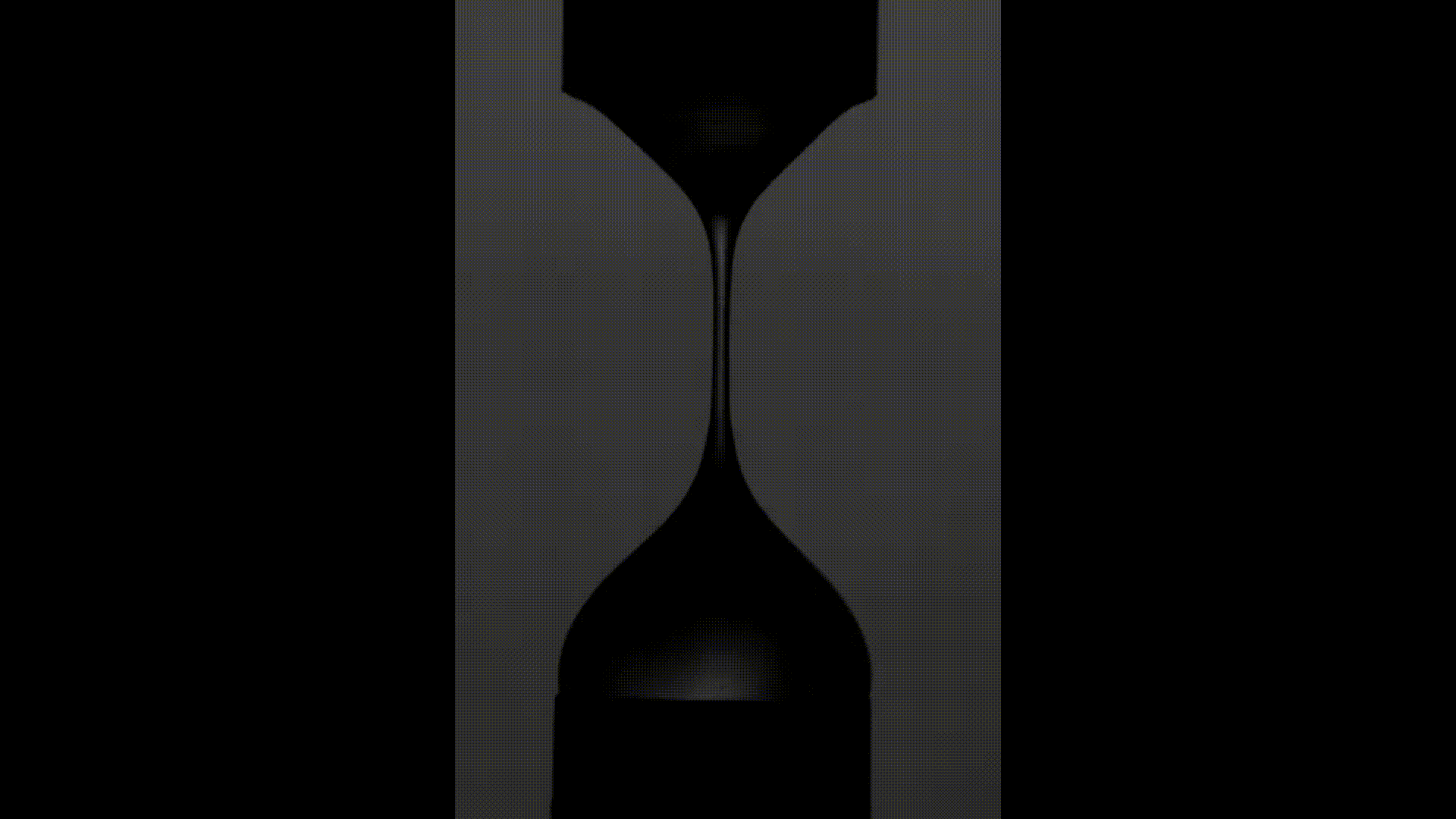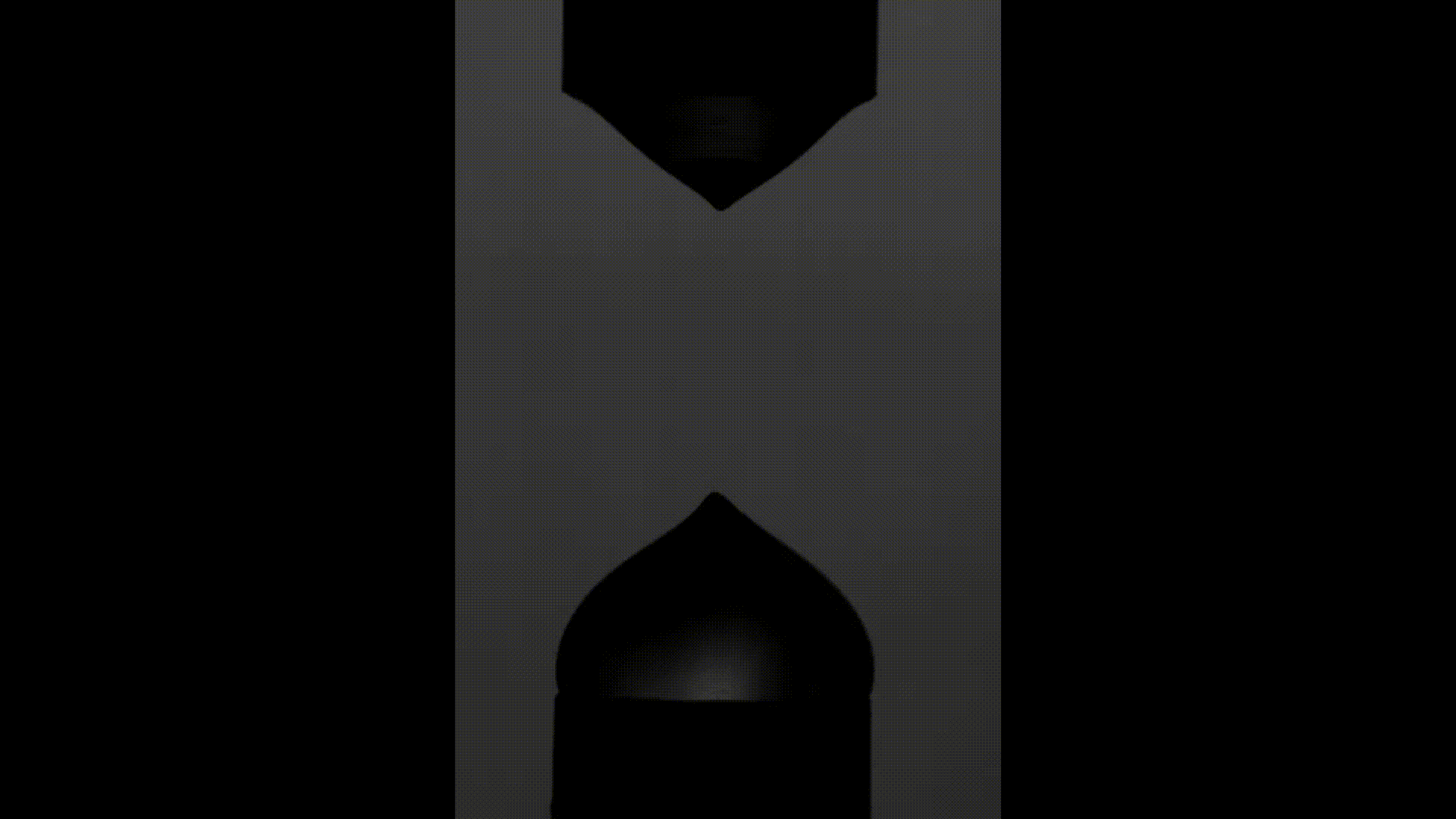
The above videos show the time evolution of two different hand sanitiser samples as they are stretched. This reflects their extensional rheology and mimics the behaviour of sanitisers in real life, for example, as we separate our hands after using them. The first sample (left) was thickened using a microgel, the second (right) using a linear polymer. The second (right hand) sample takes approximately 3 times longer to break, indicating that it may feel sticky to users. (Note, these videos are not in real time but are both rescaled in the same way).
To explore sample stickiness, the team used their Capillary Breakup Extensional Rheometer (CaBER), with the evolution of the samples recorded using the CaBER laser or a high-speed camera to create a video for analysis purposes. The two videos above are taken from these experiments. The formulation made using the linear polymer additive can be seen to take almost 3 times longer to break. Accordingly, this formulation did not fully pass the non-sticky criteria and therefore may have a less than optimal hand feel.
This work demonstrates how soft matter physics can create a framework to guide the design of formulations with desirable properties. To explore how ECFP can help with your formulation challenges, get in touch with our team.
Please get in contact with us to find out more about ECFP and whether we can help you.

An experimental approach to systematically explore the link between particle polydispersity and the structure thin-film colloidal crystals.

A new paper led by Dr Raj Tadi from the Soft Matter & Biological Physics group at the University of Edinburgh sheds light on how to control and manipulate compositional ripening in emulsions.

ECFP researchers have revealed how friction between microscopic cement grains can prevent the easy flow of fresh, unset concrete. Improving our understanding of these microscale processes will guide efforts to replace cement with more sustainable substitutes.